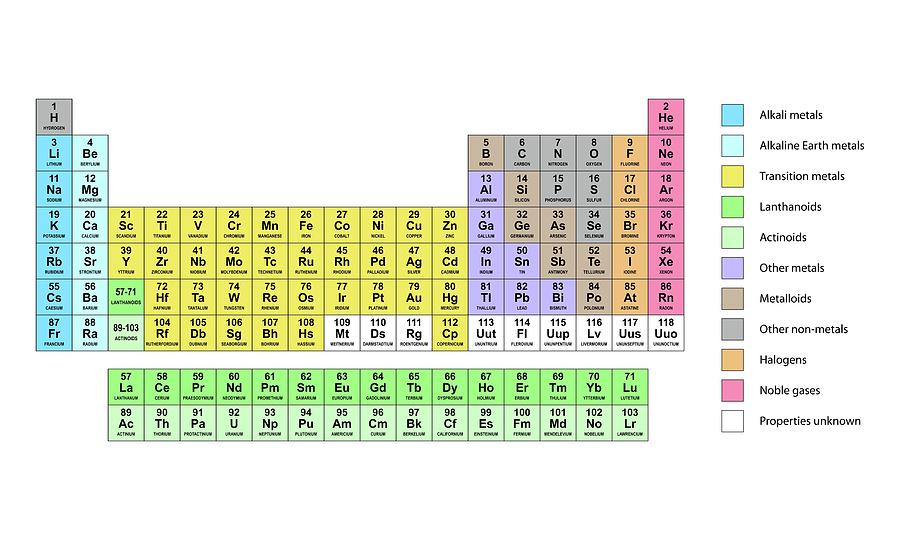
Student would then color the group to match the key made. Student told to make a key for each important group. It can also be broadly divided into metals, nonmetals, and metalloids(semimetals). Quickly reviewed the different groups on the periodic table. Note that each of these entries corresponds to one of the twelve periods respectively.\): The periodic table is split into seven periods(rows) with the inner transition metals shown separately on the bottom to decrease the width of the periodic table. :max_bytes(150000):strip_icc()/GettyImages-685863936-43a77bb814b4447490e9f2e509db980c.jpg)
In addition to the elements name, symbol, and atomic number, each element box has a drawing of one of the elements main human uses or natural occurrences. English: This pictorial periodic table is colorful, boring, and packed with information. Label the periods with 1-7 at the left of each row. Label the groups with 1-18 at the top of each column. Group six (R H superscript two, R 0 superscript three) contains the following information: (no entry for period 1), O = 16, S = 32, C r = 52, S o = 78, M o = 96, T o = 125, -, -, W = 184, -, U = 240. The Periodic Table of the Elements in Pictures.pdf. Name Period Color-Coded Periodic Table Activity Directions: Be sure to follow all instructions carefully and completely Use your textbook and any other resources to help you complete the periodic table. Note that each of these entries corresponds to one of the twelve periods respectively. Group five (R H superscript two, R superscript two 0 superscript five) contains the following information: (no entry for period 1), N = 14, P = 31, V = 51, A s = 75, N b = 94, S b = 122, -, -, T a = 182, B l = 208,. There is no set in stone standard for CPK color schemes, but nearly all molecule kits use the same common CPK colors. Note that each of these entries corresponds to one of the twelve periods respectively. Halogens green starting light green for fluorine and getting darker as you move down the group. Group four (RH superscript four, R0 superscript eight) contains the following information: (no entry for period 1), C = 12, B i = 28, T i = 48, - = 72, Z r = 90, S n = 118, ? C o = 140, ? L a = 180, P b = 207, T h = 231. Group three (-, R superscript one 0 superscript nine) contains the information: (no entry for period 1), B = 11, A l = 27, 8. Note the ach of these entries corresponds to one of the twelve periods respectively. The second group (-, R 0) contains the following information: (not entry for period 1) B o = 9, 4, M g = 24, C a = 40, Z n = 65, S r = 87, C d = 112, B a = 187, -, -, H g = 200,. Most periodic tables are color-coded to provide supplementary information about elements and item groups. Note that each of these entries corresponds to one of the twelve periods respectively. Most periodic tables are color-coded on provide added request about elements and element groups. Figure B shows the first periodic table developed by Mendeleev, which had eight groups and twelve periods. (credit a: modification of work by Serge Lachinov credit b: modification of work by “Den fjättrade ankan”/Wikimedia Commons) Figure A shows a photograph of Dimitri Mendeleev. Help students learn chemical elements with this helpful color-coded periodic table Its notebook-sized and 3-hole punched for easy reference, and includes element names, symbols, atomic numbers, electron configurations, and chemical properties. \): (a) Dimitri Mendeleev is widely credited with creating (b) the first periodic table of the elements.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
